
New regulations for aesthetics 2025: licensing, training, premises and UK compliance timeline. What aesthetics clinics must do before licensing opens.
If you're running an aesthetics clinic in England, the new regulations for aesthetics 2025 directly affect your licence to practise. Are you compliant? Two changes are already reshaping the industry: face-to-face prescribing rules already in force, and a mandatory licensing framework confirmed for rollout. This guide covers what changed, who it affects, and what your clinic must do next.
The Licensing of Non-Surgical Cosmetic Procedures framework was confirmed on 7 August 2025 (source: gov.uk). If you run an aesthetics clinic, work as an independent practitioner, or offer injectable treatments of any kind, what happens next affects your licence to practise.
Important note: This article provides general guidance on publicly announced regulatory changes. It is not legal advice. Always check current guidance from gov.uk and consult a qualified legal adviser or your professional body for advice specific to your situation.
What you'll learn:
- What the new regulations for aesthetics 2025 actually say (and what they don't yet cover)
- Which procedures will require a licence in England
- Training, insurance, and premises requirements
- The implementation timeline and what it means for your clinic in 2026
- A practical compliance checklist you can work through this week
What Changed: The 2025 Aesthetics Regulations Explained
If you're thinking "I've heard about new aesthetics regulations before and nothing changed" — you're right to be sceptical. There have been years of consultations, proposals, and false starts. This time is different.
The UK's approach to aesthetics regulation has historically been fragmented. Anyone could legally give botulinum toxin, dermal fillers, and other non-surgical treatments without formal qualifications, a licence, or any oversight. The new regulations for aesthetics 2025 change that — in two stages.
On 7 August 2025, the UK Government published its consultation response confirming plans to introduce a mandatory licensing system for non-surgical cosmetic procedures in England (source: gov.uk). From June 2025, prescription-only cosmetic treatments such as anti-wrinkle injections require a face-to-face consultation before treatment, ending the previous practice of online prescriptions for injectables.
Two regulatory changes are in motion:
Prescribing rules (in effect from June 2025): Nurses and other prescribers can no longer remotely prescribe botulinum toxin. Face-to-face consultations are mandatory before any injectable treatment (source: NMC; Acquisition Aesthetics).
Licensing framework (August 2025, implementation in progress): A new licensing system will require practitioners and premises to hold licences before offering higher-risk procedures. Secondary legislation and a phased rollout are expected to follow.
The distinction matters. The prescribing rule is already law. The licensing framework is confirmed but not yet fully implemented. Both affect your clinic in 2026 and going forward.
In short: Two changes, two timescales. Act on the prescribing rules immediately. Prepare for licensing before the application opens.
The Licensing of Non-Surgical Cosmetic Procedures Bill
So what does the licensing framework actually say? Here's the structure.
The Licensing of Non-Surgical Cosmetic Procedures framework was not a standalone bill. It was introduced through the Health and Care Act (source: House of Commons Library). That Act gave ministers powers to create a licensing system for cosmetic procedures in England. The August 2025 consultation response set out how those powers will be used.
What the consultation confirmed:
- A mandatory licensing system for non-surgical cosmetic procedures
- A risk-based categorisation of procedures (red, amber, green)
- Requirements for practitioners and premises to hold licences
- A role for local authorities in licensing oversight
The risk categories (proposed framework):
| Category | Risk Level | Examples | Likely Requirement |
|---|---|---|---|
| Red | Highest risk | Botulinum toxin, dermal fillers, chemical peels (high-strength) | Practitioner licence + premises licence |
| Amber | Moderate risk | Microneedling, semi-permanent make-up, laser treatments | Practitioner or premises licence |
| Green | Lower risk | Lash extensions, non-invasive facials | Existing local authority oversight |
Note: These categories are approximate — final category assignments may vary from the proposed framework. The classifications above reflect the House of Commons Library research briefing (September 2025). Check gov.uk for confirmed category assignments.
For example, a clinic doing anti-wrinkle injections and fillers would need both a practitioner licence and a premises licence. A lash bar doing only non-injectable treatments would likely fall into the green category — no new licence needed, existing oversight applies.
Scotland is developing its own rules after a public consultation. Separate guidance will apply north of the border (source: Medical Defense Society).
Who Needs a Licence and What It Covers
The practical question: does this apply to you?
If you perform higher-risk non-surgical cosmetic procedures in England, you will almost certainly need a licence. The framework applies separately to practitioners and to premises.
Who will need a practitioner licence:
- Anyone administering botulinum toxin injections (anti-wrinkle treatments)
- Anyone performing dermal filler procedures
- Those offering other treatments likely to fall within red or amber categories
Who will need a premises licence:
- Clinics, salons, and beauty studios where red or amber category procedures take place
- Home-based practitioners offering higher-risk treatments
This dual-licensing approach matters. If you hold a practitioner licence, the premises you work from must also be licenced. If you rent a room in another salon or visit clients at home, this affects how you structure your practice.
For example, a practitioner renting a room in a hair salon two days a week needs their own practitioner licence. The hair salon also needs a premises licence for those procedures. The duty is shared — but both licences must be in place.
Key point: Practitioner licence + premises licence. Both required. Neither covers the other.
What the licence will not resolve:
A licence is an entry requirement, not a quality guarantee. If you see compliance as just a box-ticking exercise, you're missing the point. The framework creates a minimum standard below which no practitioner can legally operate. Your reputation — and your clients' safety — depends on exceeding that standard, not just meeting it.
Ask yourself: Would I feel confident that my clinic meets the new regulations for aesthetics 2025 if a regulator visited this week? If not, the compliance audit in Section 7 is your starting point.
For a full picture of running a compliant aesthetics business, see our guide to aesthetics regulations UK.
Training and Qualification Requirements
Beyond the licence itself, qualifications are where many practitioners face the hardest questions. Here's what you need to know.
The biggest practical impact for many practitioners is what the new framework means for who can legally perform injectable treatments. Before June 2025, the route to practice was relatively open. That has changed for prescription-only treatments.
Current requirements (in effect):
- Botulinum toxin and prescription-strength fillers: From June 2025, these must be prescribed after a face-to-face consultation by a registered prescriber (nurse, doctor, dentist, or pharmacist). Online prescriptions are no longer permitted (source: NMC).
- Non-prescribers: If you are not a registered prescriber, you need a prescribing colleague or a prescribing partnership — you cannot self-prescribe injectables.
Expected qualification requirements under the licensing framework:
Practitioners seeking licences will need to demonstrate relevant qualifications. The specific benchmarks are still being developed. But two professional bodies have been central to the process: the Joint Council for Cosmetic Practitioners (JCCP) and the Cosmetic Practice Standards Authority (CPSA).
The JCCP has published a Register of Cosmetic Practitioners — those listed have voluntarily shown they meet defined competency standards (source: JCCP). Registration was voluntary before the new framework. It is increasingly seen as strong evidence of readiness for the formal licensing process.
If you're practising as an aesthetics professional:
- If you're already on the JCCP register or hold a Level 7 qualification (or equivalent) in aesthetics, you are well-positioned for the licensing process
- If you gained qualifications through short courses without any healthcare training, this is the time to check whether your credentials will meet the licensing requirements
- Training providers are updating their courses ahead of the formal standards — if you're completing training in 2026, choose a provider aligned with JCCP or CPSA
For example, a practitioner with a one-day botox course and no healthcare training is in a very different position to a nurse with a Level 7 aesthetics diploma. The framework is expected to reflect that gap.
If your qualifications are light, acting in advance of the deadline rather than waiting is the right call.
If you're only checking your qualifications when the application opens, you'll always be behind those who've been building their credentials steadily. That's not where you want to be when a deadline arrives.
For practitioners thinking of starting an aesthetics business, our guide to how to start an aesthetics business covers the business side of setting up compliantly.
Insurance and Premises Requirements
Beyond qualifications, insurance and premises are where many clinics have gaps. These affect your legal compliance and your ability to practise safely.
Insurance is not new to the aesthetics industry. But the new regulations for aesthetics 2025 affect both what cover you need and what your insurer will require.
Insurance — what's changing:
Most specialist aesthetics insurers have updated their policies since June 2025. If you are a non-prescriber doing injectables without a valid prescriber in place, your insurer may not cover you. That's not a small risk. It's the difference between a covered claim and a serious personal liability.
Check this week: Review your current policy. Does it cover the procedures you perform under the new prescribing rules? Does your insurer require evidence of a valid prescribing arrangement?
Key insurance considerations under the 2025 framework:
- Public liability insurance remains essential for all practitioners
- Treatment liability insurance must cover the specific procedures you perform — category assignments will likely affect what your policy needs
- Indemnity for prescribing: Non-prescribers should have written agreements with their prescriber, and their insurance should reflect this
Premises requirements:
Forthcoming regulations will set consistent standards for premises where procedures are performed (source: NHBF). The specific details are still being developed, but the direction is clear:
- Premises will need to meet hygiene and infection control standards
- Clinical waste disposal requirements will apply
- Emergency equipment and protocols may be required for higher-risk treatments
- Home-based and mobile practitioners will need to demonstrate that their working environment meets the required standard
If your clinic follows infection control standards and keeps treatment records, you're ahead of practitioners who have been operating informally. If you can't tell whether your premises meet the required standard, that's usually a sign you need a formal review before the application opens.
For example, a beauty studio practitioner with a dedicated treatment room, documented cleaning protocols, and locked storage for prescription products is well-positioned. Someone administering treatments at a kitchen table is not. The former will meet premises standards with minor adjustments; the latter will need significant changes or a different working arrangement.
For a broader safety view, our guide to health and safety for beauty salons covers the safety basics relevant across the beauty salon industry.
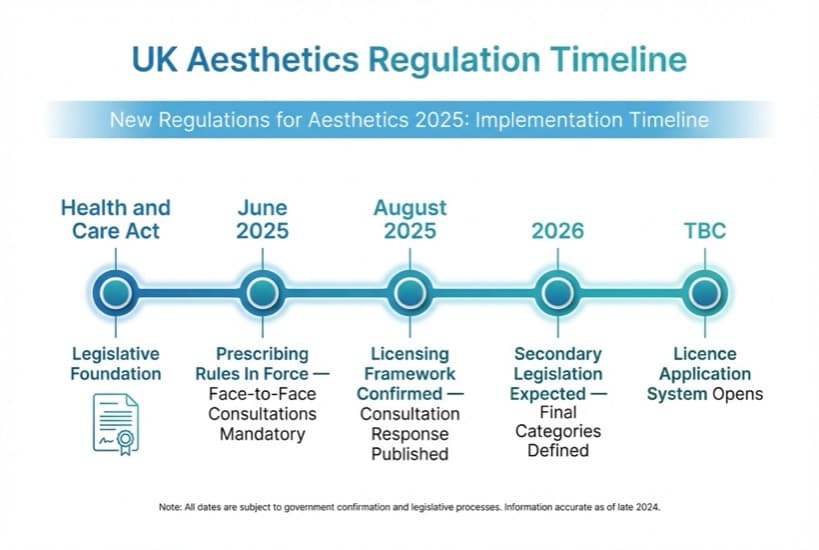
UK aesthetics regulation timeline: key dates and milestones
Timeline: What You Need to Do and When
Understanding the regulations is one thing. Knowing when to act is another.
The implementation is being phased. Major regulatory changes require secondary legislation, system infrastructure, and a transition period for practitioners to become compliant. Here's where things stand.
What we know about the timeline:
| Date | What Happened / What's Expected |
|---|---|
| Historical 2022 | Health and Care Act passed — powers granted to create licensing system |
| June 2025 | Face-to-face consultation rules for prescription-only injectables came into force |
| 7 August 2025 | Government published consultation response confirming licensing framework |
| Late 2025 / 2026 | Secondary legislation expected to define final procedure categories and standards |
| TBC | Licence application system opens for practitioners and premises |
| TBC | Grace period for existing practitioners to obtain licences |
| TBC | Unlicensed practice for red/amber procedures becomes illegal |
March 2026 status: As of this writing, the licensing framework has been confirmed but the formal application system has not yet opened. The prescribing rules are in full force. Practitioners who prepare in advance — before the formal application opens — will have the smoothest path through licensing. Those with strong qualifications, proper insurance, and compliant premises are already well-positioned.
If you're reading this thinking "I don't have time to deal with this" — you're not alone. Many independent aesthetics practitioners are juggling a full treatment schedule alongside the admin burden of running their own business. Leaving compliance until the deadline — that never works (source: NHBF compliance guidance). The deadline may arrive faster than you expect.
The cautionary reality: If you're only looking at the new regulations for aesthetics 2025 when the licence application opens you'll always be scrambling to catch up against practitioners who prepared early. Those who've been operating informally — weekend course qualifications, no prescriber arrangement, makeshift treatment rooms — will find themselves unable to meet the criteria quickly. That's not a small cohort of the industry.
Why this matters: The practitioners who will struggle most with the new regulations for aesthetics 2025 are those who delayed preparation. The licensing application process — when it opens — will not be a quick form-fill. It will require documented evidence of qualifications, prescribing arrangements, insurance, and premises standards. Building that evidence takes time.
Knowing the timeline is useful. But it's not a reason to wait. Every month of preparation before the licence application opens is one month ahead of those who aren't acting.
For example, a clinic that starts gathering qualification records and insurance documentation in March 2026 will be well-prepared when the licence application opens — whatever that date turns out to be. A clinic that waits will be scrambling.
For a broader view of building a compliant aesthetics business, see our aesthetics business hub.
Check Your Compliance This Week
You don't need to wait for the formal licensing application to begin preparing under the new regulations for aesthetics 2025. Start with this structured audit — get the basics right and improve your compliance position before licensing opens:
This week, audit your aesthetics practice compliance:
Day 1 to 2: Check your prescribing arrangements
- Are you a registered prescriber? If not, do you have a written agreement with one?
- Is your prescriber aware of and compliant with the June 2025 face-to-face requirements?
- Review your insurance policy — does it reflect your current prescribing arrangement?
Day 3 to 4: Review your qualifications
- List every qualification you hold relevant to the treatments you perform
- Check whether your training provider is aligned with JCCP or CPSA frameworks
- Visit the JCCP register (jccp.org.uk) — if you're eligible to apply, consider registering in advance of the licensing launch
Day 5 to 7: Assess your premises and documentation
- Does your treatment space meet clinical hygiene standards?
- Do you have documented infection control procedures?
- Are your client consent forms and treatment records up to date and stored appropriately?
For example, a typical compliance gap found during audits: a practitioner has a face-to-face consultation policy in place but hasn't updated their client consent form to reflect the June 2025 requirements. That's a fixable gap — but only if you know it exists.
If you only have 30 minutes a week:
Your minimum viable compliance action this week:
- Day 1 to 2: Check your insurance policy is current and covers your procedures under the new prescribing rules
- Day 3 to 4: Verify your prescribing arrangement meets the June 2025 face-to-face requirement
- Day 5 to 7: Bookmark gov.uk ("licensing non-surgical cosmetic procedures") and the JCCP register for regular updates
Where to find official guidance:
- gov.uk — Search "licensing non-surgical cosmetic procedures" for the consultation response and March 2026 updates
- JCCP (jccp.org.uk) — Practitioner register and competency frameworks
- NHBF (nhbf.co.uk) — Guidance for salons and beauty businesses
- CPSA (cosmeticpractice.org.uk) — Standards authority for cosmetic practice
For the marketing side of running a compliant aesthetics clinic, discover how to build trust with clients in a regulated environment in our aesthetics clinic marketing guide. You can also explore aesthetics marketing strategies and aesthetics marketing ideas for more practical approaches.
Frequently Asked Questions
What is the new aesthetic law 2025?
The "new aesthetic law 2025" covers two linked changes. First, the June 2025 rule: face-to-face consultations are required before prescription-only injectable treatments. Second, the August 2025 government confirmation: a mandatory licensing system for non-surgical cosmetic procedures in England. The licensing framework is being rolled out in phases through 2026.
What are the new aesthetic regulations for 2026?
In 2026, the key development is secondary legislation to define final licensing categories and the formal application process. Practitioners should already be preparing — reviewing qualifications, prescribing arrangements, insurance, and premises — before the formal licence application opens.
What are the new aesthetic rules in the UK?
The key rules in force since June 2025: prescription-only cosmetic treatments (including botulinum toxin) require a face-to-face consultation before treatment — online prescriptions are no longer permitted. A mandatory licensing system for higher-risk non-surgical procedures is confirmed for England. The new regulations for aesthetics 2025 form the basis of this framework.
Who is exempt from the new aesthetics licensing rules?
The licensing framework focuses on higher-risk non-surgical procedures in the red and amber categories. Lower-risk treatments are likely to stay under existing local authority oversight. Medical professionals in regulated healthcare settings operate under separate rules. Check the JCCP and gov.uk for updates on exemptions through 2026.
For example, a lash technician offering lash extensions, tints, and brow shaping would likely fall into the green category — no new licence required under the current proposed framework. A nurse aesthetician offering botulinum toxin and dermal fillers would need both a practitioner licence and compliant premises.
Does the new regulations for aesthetics 2025 framework apply in Scotland and Wales?
No. The new regulations for aesthetics 2025 (confirmed August 2025) apply to England only. Scotland is developing its own rules after a separate public consultation. Wales and Northern Ireland follow their own devolved processes. If you practise across borders, check the guidance for each nation.
Whether you're working with an aesthetics marketing agency or managing marketing yourself, understanding these regulations is essential. For practitioners exploring the business side, see our guides on medical aesthetics marketing and creating an aesthetics business plan.
This article covers the new regulations for aesthetics 2025 as publicly announced and confirmed at the time of writing. Regulations and implementation timelines may have been updated since publication. Always check current guidance at gov.uk and consult a qualified legal adviser for advice specific to your circumstances.
For UK aesthetics practitioners
Stay Ahead of Aesthetics Regulations
Local Brand Hub helps UK aesthetics clinics manage their marketing while staying compliant. Build trust, attract clients, and keep your online presence up to date.
Try It FreeKey Takeaway
The new regulations for aesthetics 2025 mark a genuine shift — not another consultation that fades away. The prescribing rule is already law (face-to-face consultations mandatory since June 2025). The licensing framework is confirmed but not yet fully implemented. Both practitioners and premises will need licences for higher-risk procedures. Your qualifications, insurance, and premises all need to be in order before the application opens. Scotland, Wales, and Northern Ireland have separate processes. For most practitioners who've been running their clinic properly, this regulation confirms their position rather than disrupts it.
About the Author
Local Brand Hub
Empowering UK Businesses
Local Brand Hub provides comprehensive business management tools designed specifically for UK local businesses to streamline operations, automate marketing, and grow revenue.
More articlesRelated Articles
 Industry Insights
Industry InsightsBarista Training Cornwall: SW England Local Routes
Barista training Cornwall guide — Origin Coffee school, Truro and Falmouth options, distance learning blends, seasonal cafe staffing.
 Industry Insights
Industry InsightsBarista Training Dublin: Republic of Ireland Guide
Barista training Dublin: SCA Ireland courses, roastery schools (3fe, Cloud Picker), and what UK baristas need to know about RoI travel.
 Industry Insights
Industry InsightsBarista Training Northern Ireland: Belfast & Beyond
Barista training Northern Ireland — Belfast SCA-authorised trainers, NIHF routes, cross-border options with the Republic of Ireland.